- Break down the most common misconceptions in Class 10 Chemistry (CBSE-focused but useful for all boards)
- Show how board exam questions test these misconceptions
- Give correction rules you can apply immediately
- Provide a structured daily strategy to fix them permanently
Misconception 1: “Chemical Reactions Are Just for Memorizing”

Why Students Get Confused
In Chemical Reactions and Equations, many students focus only on memorizing equations:- Zn + HCl → ZnCl₂ + H₂
- CaCO₃ → CaO + CO₂
- NaOH + HCl → NaCl + H₂O
- Identify the type of reaction.
- Explain oxidation and reduction.
- Predict products.
- Justify why a reaction occurs.
The Conceptual Correction Rule
For every reaction, answer three questions:- What type of reaction is this?
- What changed chemically?
- Why did it happen?
Board-Style Tricky Example
Question:
Why is respiration considered an exothermic reaction?Most students write: “Because energy is released.”
Incomplete.
Correct explanation:In respiration, glucose reacts with oxygen to form carbon dioxide and water, releasing energy. Since heat energy is released during the reaction, it is classified as exothermic.Board exams often test classification + reasoning together.Misconception 2: “Balancing Equations Is Just a Formal Step”
The Real Problem
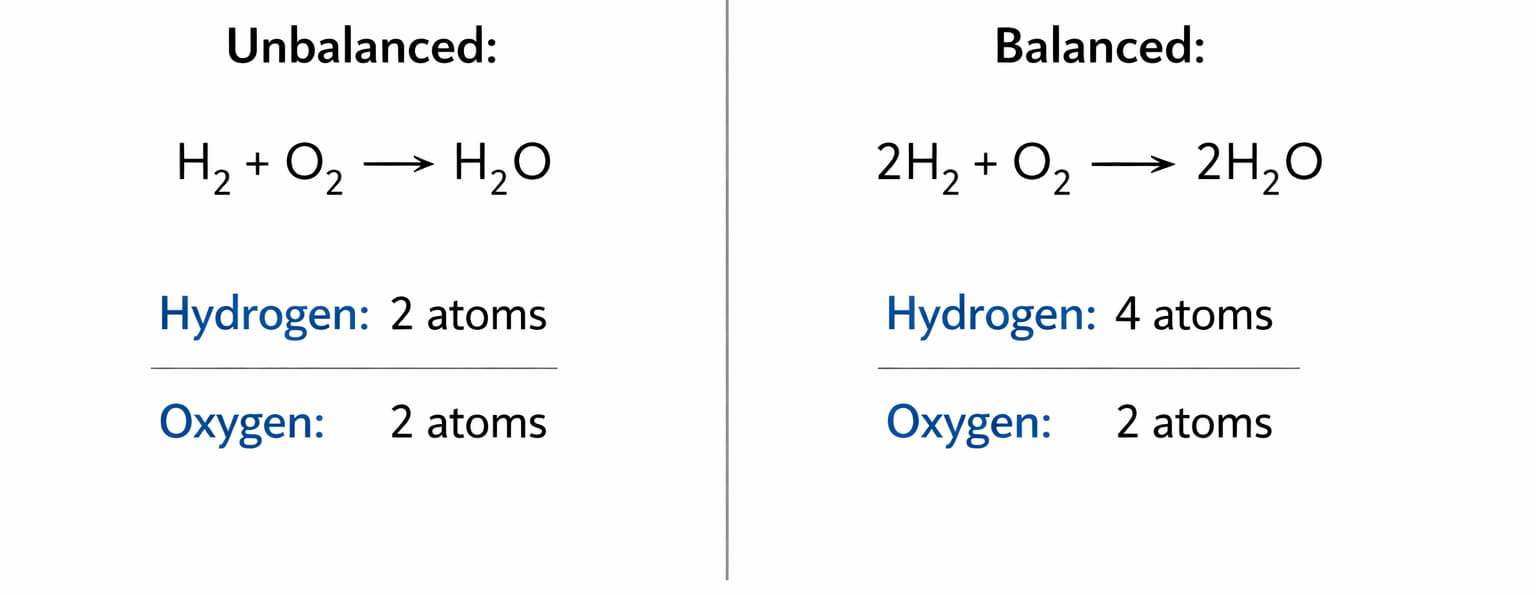
Students treat balancing like a math adjustment instead of a chemical principle.They forget the Law of Conservation of Mass.Unbalanced: H₂ + O₂ → H₂OBalanced: 2H₂ + O₂ → 2H₂OBut why 2?Because total hydrogen atoms must be equal on both sides.Deeper Understanding
Balancing ensures:Total number of atoms of each element on LHS = RHS.This principle later connects to mole concept in Class 11. If balancing is weak now, stoichiometry becomes difficult later.Board-Style Tricky Example
Question:
Balance the equation and identify the type of reaction: Fe + H₂O → Fe₃O₄ + H₂Correct balanced equation: 3Fe + 4H₂O → Fe₃O₄ + 4H₂Type: Redox reaction (iron is oxidized, hydrogen is reduced).Students often balance incorrectly or fail to identify redox nature.Misconception 3: “Strong Acid Means Concentrated Acid”
This is one of the most common Class 10 Chemistry mistakes.The Confusion
Students mix up:- Strong vs weak
- Concentrated vs dilute
Clear Definitions
- Strong acid → Completely ionizes in water (e.g., HCl)
- Weak acid → Partially ionizes (e.g., CH₃COOH)
- Concentrated → High amount of solute
- Dilute → Low amount of solute
Deeper Insight (Often Ignored)
Strength depends on ionization ability, not amount present.For example:0.1M HCl may have more H⁺ ions than 1M acetic acid because HCl ionizes completely.Understanding this prevents confusion in pH-related questions.Misconception 4: “Physical and Chemical Changes Are Easy to Identify”
Students assume:- Change in shape = physical
- Change in color = chemical
Correct Rule
A chemical change forms a new substance with new properties.Example:- Rusting of iron → Chemical change
- Melting of ice → Physical change
But what about:
- Burning magnesium ribbon?
- Digestion of food?
- Dissolving ammonium chloride in water?
- Is a new substance formed?
- Is the change easily reversible?
Board-Style Tricky Example
Question:
When copper sulphate solution reacts with iron nails, the solution turns green. Is this a physical or chemical change? Why?Correct Answer:It is a chemical change because iron displaces copper from copper sulphate, forming iron sulphate (green) and copper metal. A new substance is formed.Many students only write “color change,” which is incomplete reasoning.Misconception 5: “Reactivity Series Is Just for Memorizing Order”
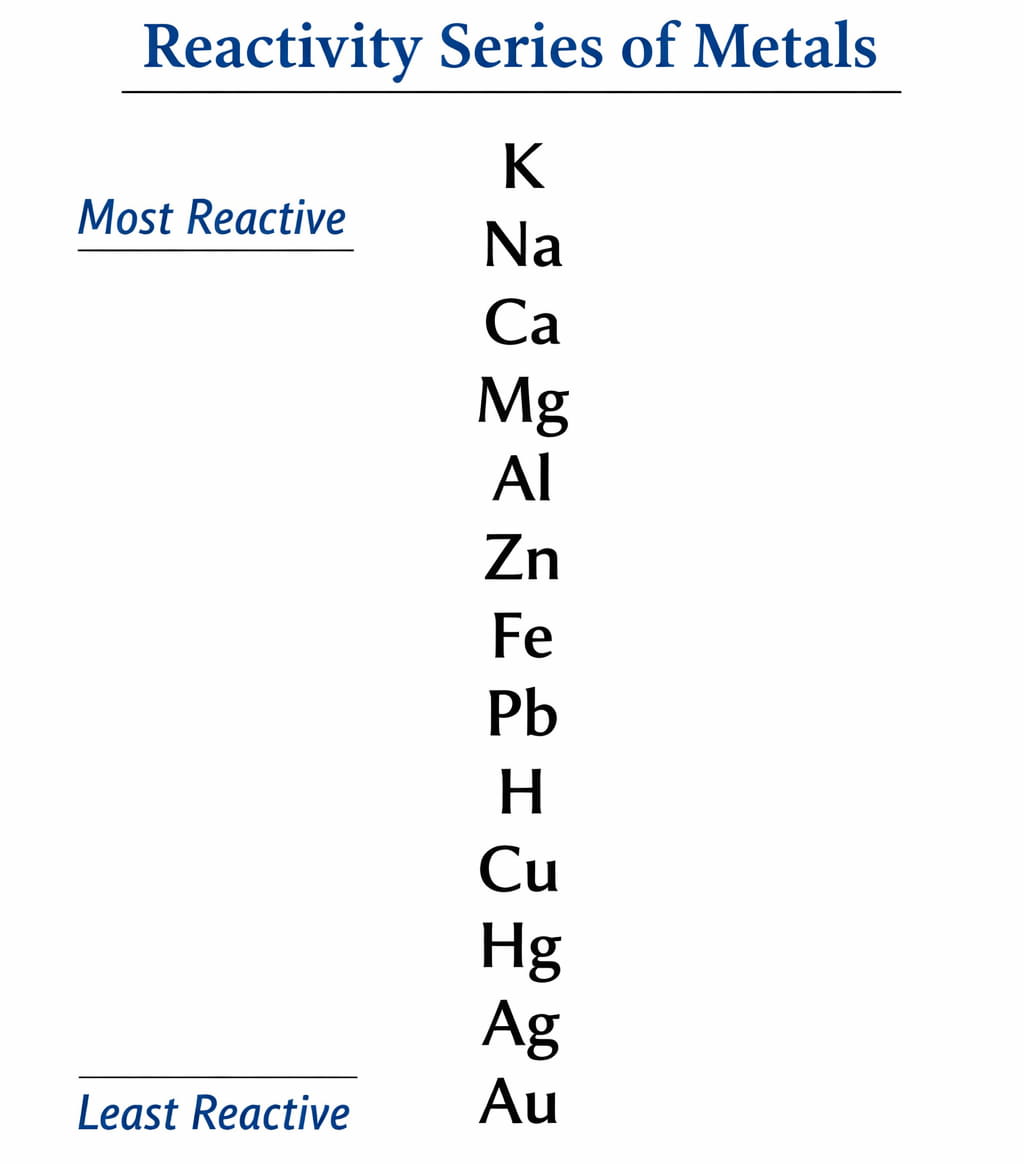 Students memorize:K > Na > Ca > Mg > Al > Zn > Fe > Pb > H > Cu > Hg > Ag > AuBut cannot apply it.
Students memorize:K > Na > Ca > Mg > Al > Zn > Fe > Pb > H > Cu > Hg > Ag > AuBut cannot apply it.Application Rule
A more reactive metal displaces a less reactive metal from its salt solution.Example:Zn + CuSO₄ → ZnSO₄ + CuBut:Cu + ZnSO₄ → No reactionIf hydrogen is above a metal in the reactivity series, that metal can displace hydrogen from acids.This logic is frequently tested in 2–3 mark board questions.Misconception 6: “All Compounds Conduct Electricity”
In Carbon and Its Compounds, students often assume all substances conduct electricity.Correct Understanding
- Ionic compounds conduct electricity in molten or aqueous state.
- Covalent compounds generally do not conduct electricity because they do not form ions.
A Unique Insight Most Students Ignore
Here is something rarely discussed:Most misconceptions are not due to difficult concepts. They are due to passive revision.Students:
- Read notes repeatedly
- Highlight textbook lines
- Watch explanation videos
- Writing balanced equations without seeing solutions
- Explaining reactions aloud
- Solving mixed questions
- Analyzing wrong answers
Practical 60-Minute Daily Strategy to Fix Misconceptions
Step 1: Concept Review (15 minutes)
- Study one small subtopic.
- Write 5 key points from memory.
Step 2: Mixed Question Practice (30 minutes)
- 15–20 questions:
- 5 theory
- 5 reasoning
- 5 application
Step 3: Mistake Analysis (15 minutes)
For every wrong answer, write:- What concept was weak?
- Was it a careless mistake?
- What is the correct reasoning?
- Below 60% → Concept unclear
- 60–80% → Needs revision
- Above 85% → Strong clarity
Common Mistakes in Class 10 Chemistry (Must Avoid)
- Memorizing reactions without understanding type
- Ignoring balancing practice
- Mixing strong vs concentrated
- Avoiding numerical questions
- Not applying reactivity series logically
- Writing incomplete reasoning answers in board exams
- Skipping mistake analysis after practice
Chemistry improves through correction, not repetition.
FAQ: Common Questions Students Ask
1. What are the most common mistakes in Class 10 Chemistry?
Students commonly mix up strong vs concentrated acids, make balancing errors, misidentify reaction types, and fail to explain reasoning in board answers.2. How can I improve in Chemical Reactions and Equations?
Practice balancing daily, classify reaction types, and explain why each reaction occurs instead of memorizing blindly.3. Is memorizing the reactivity series enough for board exams?
No. You must know how to apply it to predict displacement reactions and reactions with acids.4. Why do I lose marks even when my final answer is correct?
Board exams award marks for steps and reasoning. Incomplete explanations often reduce marks.5. How many questions should I practice daily for Chemistry?
Around 15–20 mixed questions daily with proper error analysis is sufficient for steady improvement.
What You Should Do Today
- Choose one chapter (e.g., Acids, Bases and Salts).
- Solve 20 mixed questions.
- Check answers.
- Write down 3 conceptual mistakes.
- Revise those exact weak areas.
Conclusion
Class 10 Chemistry becomes difficult only when misconceptions remain uncorrected.If you:- Understand reactions instead of memorizing
- Balance equations with logic
- Apply the reactivity series correctly
- Differentiate similar terms clearly
- Practice actively and analyze mistakes
Start Learning Smarter Today
Join 60,000+ students already studying with Super Tutor
Try Super Tutor Free